|
Second order reactions are chemical reactions of which the rate of reaction depends on the molar concentration of two of the reactants or the second power of one reactant that involved in the reaction. Therefore, if we increase the concentration of reactant by 2 times, the rate of reaction increases by 2 times. What is the Difference Between First and Second Order Reactions?įirst order reactions are chemical reactions of which the rate of reaction depends on the molar concentration of one of the reactants that involved in the reaction. If there are two reactants, the rate of reaction will depend on the first power of the concentration of each reactant. Therefore, according to the above definition for order of reaction, the sum of the powers to which the reactant concentrations are raised in the rate law equation will always be 2. Therefore, from experimental determinations, the value of m is 1. Here k is the rate constant for this reaction and m is the order of the reaction. 
Since it has only one reactant, we can write the reaction and the rate equation as follows. In the decomposition reaction of N 2O 5, it forms NO 2 and O 2 gases as products. Let us consider an example in order to understand this concept. But sometimes, there are more than one reactants that take part in these reactions, then one of these reactants will determine the rate of the reaction. Then the concentration of that reactant determines the rate of the reaction. There can be either a single reactant that takes part in these reactions. Therefore, according to the above definition for the order of reaction, the sum of the powers to which the reactant concentrations are raised in the rate law equation will always be 1. 
Summary What are First Order Reactions?įirst order reactions are chemical reactions of which the rate of reaction depends on the molar concentration of one of the reactants that involved in the reaction. Side by Side Comparison – First vs Second Order Reactions in Tabular Formĥ. There are several forms of reactions according to this definition zero order reactions (these reactions does not depend on the concentration of reactants), first order reactions and second order reactions. The order of a reaction is the sum of the powers to which the reactant concentrations are raised in the rate law equation. #color(blue)(t = ln((_0)/(_0))/(k(_0 - _0)))#Įven though we are doing half-life, we don't know their half-lives, so we can't assume that # = _0/2# or that # = _0/2#.The key difference between first and second order reactions is that the rate of first order reactions depends on the first power of the reactant concentration in the rate equation whereas the rate of second order reactions depends on the second power of the concentration term in the rate equation. In the interest of time (and partial fractions is not the focus here): With this, we need to use partial fractions to integrate the right side. Using separation of variables again, we have: When the concentrations of both compounds decrease, they decrease by an unknown amount, #x#, so we have: Notice how we cannot just pick # and call it good. (Clearly if #A = B#, then we are just doing (i).) Using #_0 ne _0#, we get a second order reaction of two first-order components #A# and #B#: Then, you can integrate each side with respect to the variable in question. 
What we can do is a separation of variables: 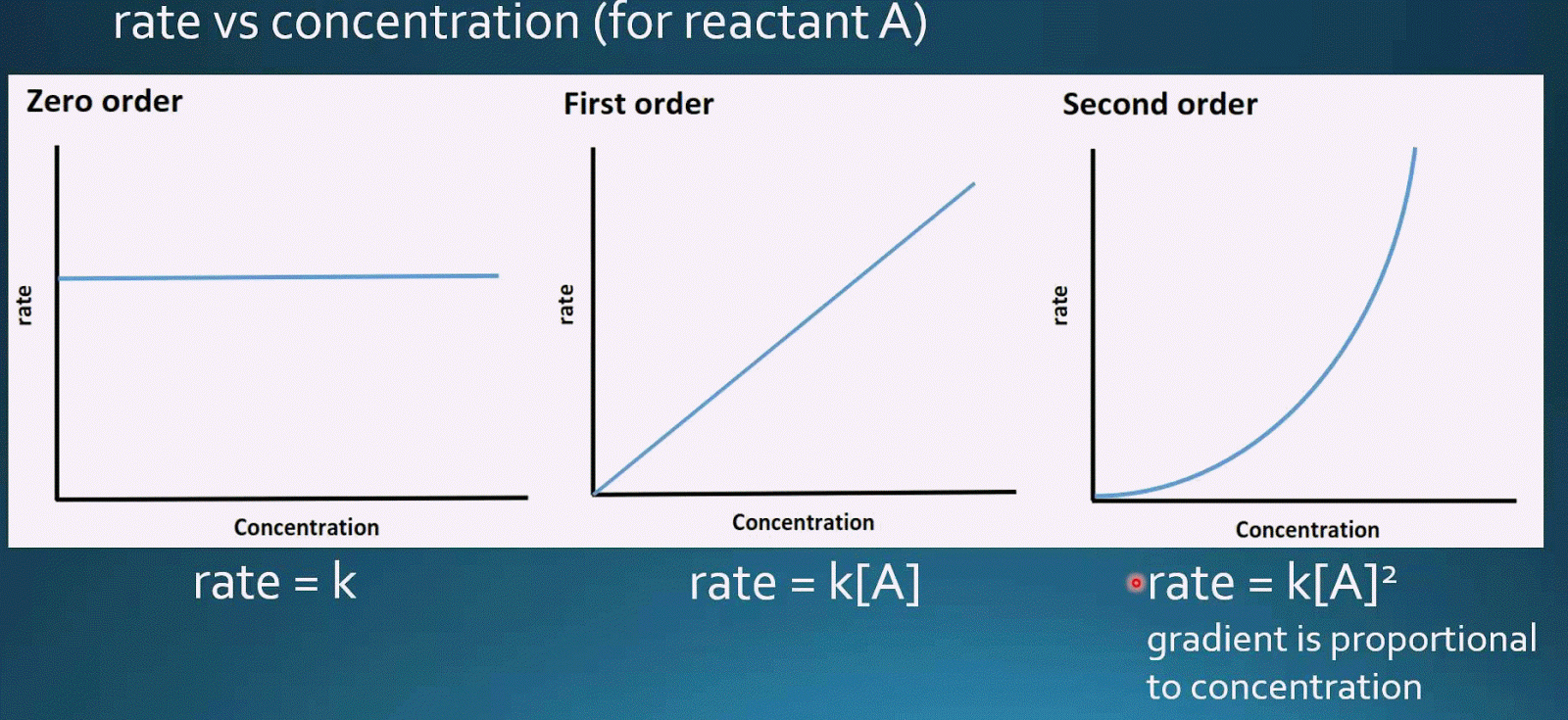
It is not two #A# reactants reacting with a #B# reactant. Note that despite the fact you have two #A# reactants, you use the stoichiometric coefficient #\mathbf1#, not #2#, because #A# is reacting with itself. With equal amounts of two disappearing reactants, and calling the appearing product #C#, you have the rate law: Only through assuming that each reaction is an elementary reaction do we have
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
